The 1st Point-of-Care (POC) Antibody Test for COVID-19
with Fingerstick Whole Blood
Authorized by FDA!
Click the link to review FDA NEWS RELEASE on Sept 23, 2020 in details: "FDA Authorizes First Point-of-Care Antibody Test for COVID-19" about Assure COVID-19 IgG/IgM Rapid Test Device.
This authorization means that Fingerstick Blood Samples can now be tested in POC settings like Doctor's offices, Urgent Care Centers, Emergency Rooms and Hospitals, rather than having to be sent to a central lab for testing.
You can find Assure COVID-19 IgM/IgG Rapid Test (as the third company listed) to review the performance data summarized by FDA on EUA Authorized Serology Test Performance, and you can also review NCI's Frederick National Laboratory for Cancer Research Evaluation Report for details.
What is COVID-19
The novel coronaviruses belong to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.

Indication
Qualitative detection and differentiation of IgM and IgG antibodies to SARS-CoV-2 in human venous whole blood (sodium EDTA), serum, or plasma (sodium EDTA). Intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection. Emergency use of this test is limited to authorized laboratories.
Warning
- This test has not been FDA cleared or approved
- This test has been authorized by FDA under an EUA for use by authorized laboratories
- This test has been authorized only for the presence of IgM and IgG antibodies against SARS-CoV-2, not for any other viruses or pathogens
- This test is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner
- Positive results may be due to past or present infection with non-SARS-CoV-2 coronavirus strains, such as coronavirus HKU1, NL63, OC43, or 229E
- This product is intended for professional use and not for home use
- Not for the screening of donated blood
Materials Provided
- 20 Individually packed test devices
- 20 Individually packed buffer
- 20 Disposable pipettes
- 20 Alcohol prep pads
- 20 Sterile safety lancets
- 1 Package insert
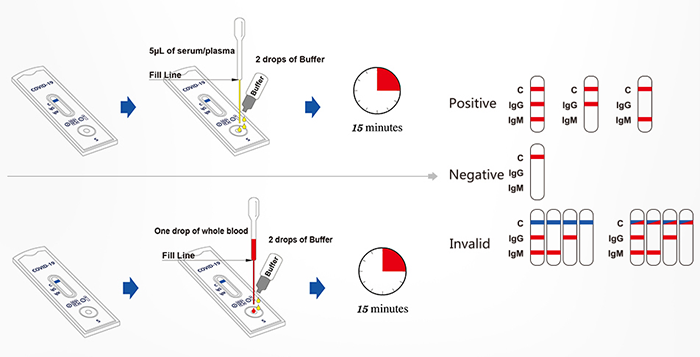
Test Procedure
- 1. Bring the pouch to room temperature before opening. Remove the test device from the sealed pouch and use it as soon as possible
- 2. Place the test device on a clean and level surface. Note: There should be a blue line in the control region (next to “C”). Discard the device if there is no blue line
- 3. Label the test with patient or control identification
- 4. Add the specimens
- 5. Wait for the blue line change to red line, read results at 15 minutes
For Serum or Plasma Specimen
Using the provided disposable pipette, draw the specimen up to the Fill Line, and transfer all the specimen (appr. 5 µL) into the specimen well of the test device, then add 2 drops of buffer and start the timer.
For Fingerstick Whole Blood Specimen
Using the provided disposable pipette, draw the specimen above the fill line (avoid the specimen entering the bubble of disposable pipette) and transfer one drop of the specimen into the specimen well of the test device, then add 2 drops of buffer and start the timer.